products

-Reactive Paint that Controls Rust with Rust-
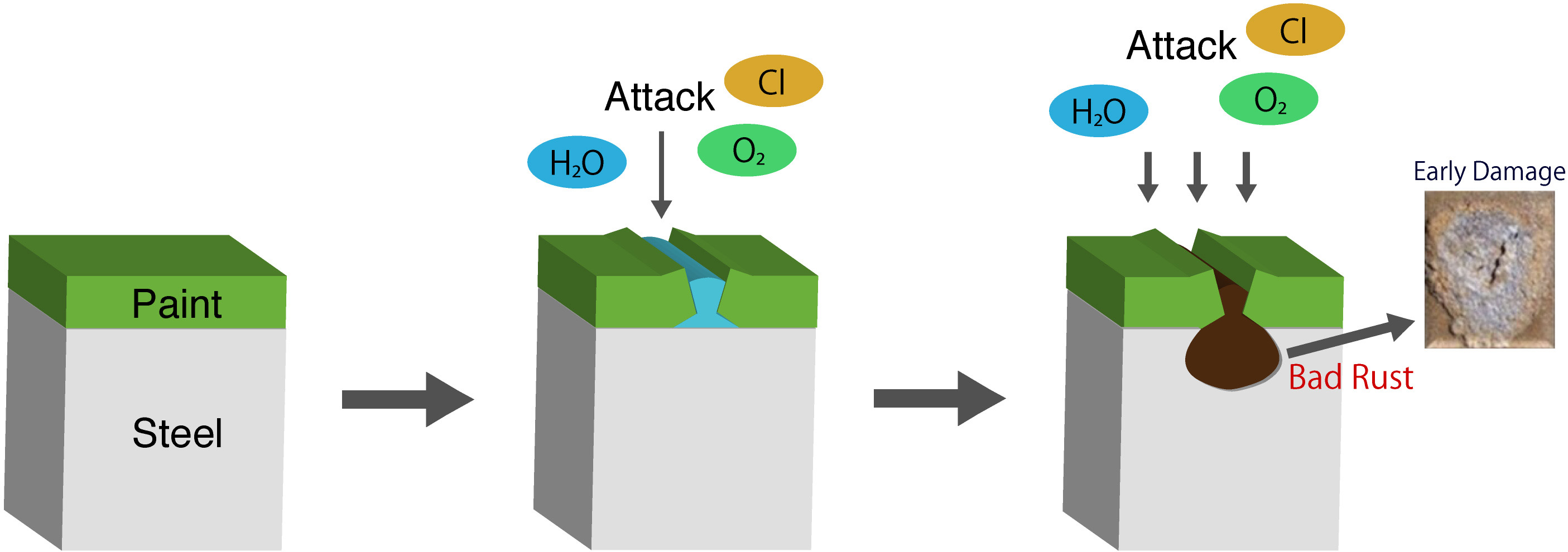
For corrosion protection of iron materials, paint is generally used to shield iron from the corrosive environment, but the corrosive environment penetrates through the paint film causing corrosion of iron. Since iron is present inside the earth’s crust in the form of iron oxide (iron ore), iron always tries to return to the form of iron ore. From the human perspective, this appears as corrosion, but from the perspective of nature iron is returning to the earth. This is known as rusting. That is, in the case of paint film that normally aims to provide an environmental shield, peeling of the paint film starts due to the corrosion of the base metals by the invasion of corrosive factors because of either paint film deterioration or pinhole defect, and repairs such as repainting is required causing the life cycle cost of steel structures to increase.
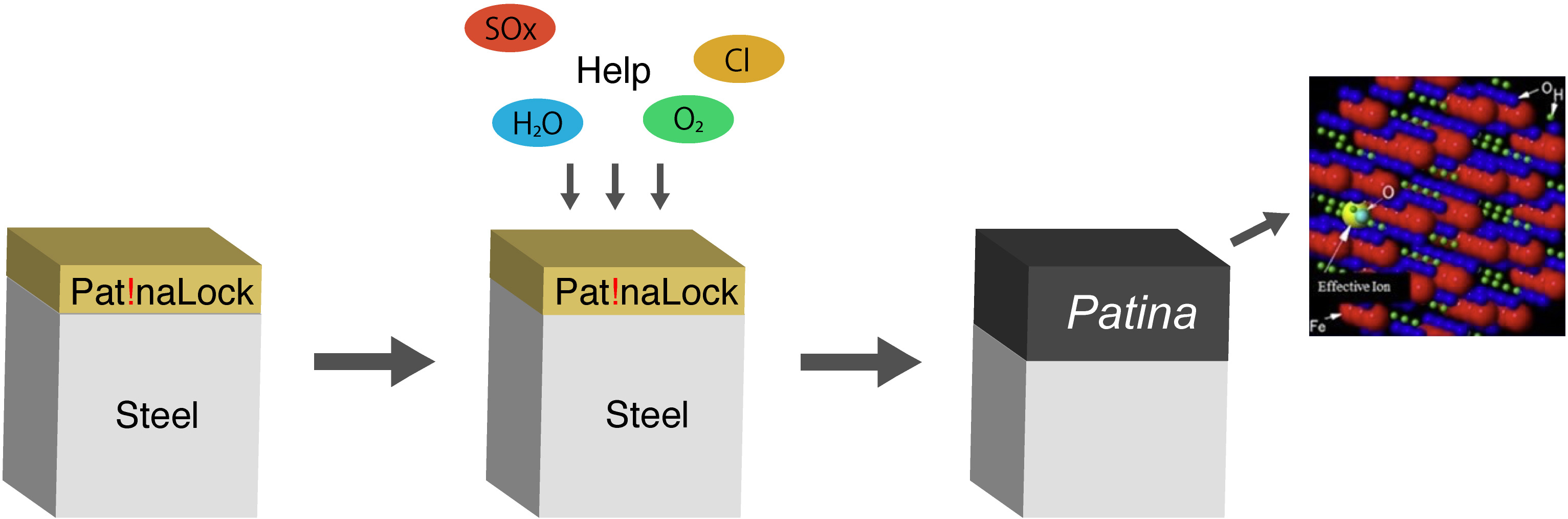
Reactive paint Pat!naLock® reacts strongly with iron and the corrosive environment creating a highly corrosion-resistant oxide layer mainly made of rust on the iron surface suppressing the corrosion of iron. In other words, Pat!naLock® harmonizes with the natural environment and returns iron surfaces to their original form that is oxide, due to which iron settle downs and does not rust, and this essentially suppresses further corrosion.
Pat!naLock® is applied to the steel surface. Corrosion is prevented from advancing by promoting the formation of an oxide layer on the iron & steel or zinc plated steel surfaces by setting the oxide layer that is formed to a state of high corrosion resistance. The reactive paint can be applied with normal methods such as spraying, brush coating, and roller coating without any special preparations.
This product can be applied to not only a wide range of newly constructed steel structures such as bridges and steel towers, and various plant facilities but also to existing structures without completely eliminating corrosion residual rust products such as red rust. The reactive paint can be applied after a hand cleaned finish of surface preparation.
Pat!naLock® acts on steel with the help of the environment rather than shielding the surface from the environment by creating the so called good rust “Patina” under all environments, significantly suppressing the corrosion of steel for a long time.
This a new technology to semi-permanently suppress the corrosion of iron by reacting with iron using the environment’s help to form corrosion resistant rust “Patina” on the surface without shielding iron from the natural environment.
Previous Products(normal coatings)
Pat!naLock®
Products
 |
Pat!naLock® -Ferric Series-This can be applied to steel materials with coating specification of 1 to 2 layers. Steel surfaces will be protected with corrosion resistant rust “Patina”. |
 |
Pat!naLock® -Galva Series-This can be applied to steel and galvanized steel with coating specification of 2 to 4 layers.This product can be matched to any color with a special overcoat. Since steel surfaces are protected with corrosion resistant rust “Patina”, life of the coating increases significantly. |
 |
Pat!naLock® -Σ Series-Reactive paint that can form an oxide layer with high corrosion resistance,which is in harmony with the environment in severe corrosive surroundings such as corrosive environment of chlorides and acidic corrosive environment such as sulfuric acid. For example, the paint effective for the acidic corrosion environment such as the exhaust gas treatment equipment of thermal power plants. Jointly developed with The Kansai Electric Power Company Inc., Mitsubishi Hitachi Power Systems Ltd., and Nagase & Co., Ltd. |
Pat!naLock® Series Common Features
Can be applied to new construction and repairs of existing construction. Can be applied with hard-cleaned finish of surface preparation for repairs.
- Please see the brochures for details.
Application Examples and Achievements
 |
 |
 |
| Transmission towers | Plant equipment | Lighting towers |
 |
 |
 |
| Creative architectural designs | Parking lots | Substation equipment |
 |
 |
 |
| Communication towers | External steel frames | Bridges |
| Pat!naLock® -Ferric Series- | Pat!naLock® -Galva Series- | Pat!naLock® -Σ Series- | |
| Applications | New construction and repair | New construction and repair | New construction and repair |
| Applicable steel | Iron | Iron, zinc plating | Iron, zinc plating |
|
Surface preparation |
Blasting or hard-cleaned finish*1 | Blasting or hard-cleaned finish*1 | Blasting or hard-cleaned finish*1 |
| Painting specification | 1 to 2 layers coating | 2 to 4 layers coating | 1 or more layer coating |
| Color choise | Restricted | Allowed (dedicated top coat) | Allowed (dedicated top coat) |
| Top coating paint | Pat!naLock®-SB *2 | Pat!naLock®-Top coat *3 | Pat!naLock®-Top coat *3 |
*1 ASK for details
*2 Pat!naLock®-Top coat (Butyral)
*3 Pat!naLock®-Top coat (fluorine, urethane/strong solvent, weak solvent)
Technical information:Example of Fundamental Research Papers
- 長期大気腐食による耐候性鋼安定さび層の安定化過程 材料と環境, 43, 26(1994).腐食防食協会 論文賞(平成8年5月)
- 耐候性鋼の最終安定さび層を構成するCr置換微細ゲ-サイトの傾斜組成分布とイオン選択性 鉄と鋼, 83, 448(1997). 日本鉄鋼協会 俵論文賞(平成10年4月)
- Long-Term Growth of Protective Rust Layer Formed on Weathering Steel by Atmospheric Corrosion during A Quarter of A Century, Corrosion Science, 36, No.2, (1994), 283-299.
- Dependence of Corrosion Potential and Corrosion Rate of a Low-Alloy Steel upon Depth of Aqueous Solution, Corrosion Science, 40, No.9, (1998), 1447-1453.
- Recent Research and Development for solving Atmospheric Corrosion Problems of Steel Industries in Japan, Hyperfine Interactions, 139/140, (2002), 153-166.
- Nanostructure of Protective Rust Layer on Weathering Steel”, Mater. Trans., 45, No.6, (2004), 1920-1924.
- 反応性塗料を塗布した発錆炭素鋼に生成するさび層の構造と防食性, 材料と環境, Vol.66, No.3, pp.93-98(2017).
- Structure and Corrosion Protection of Rust Layer Formed on Pre-rusted Carbon Steel Covered with Reactive Paint Coating, Corrosion Engineering, Vol. 66, No. 3, pp. 64–70(2017)
Winning Prizes
- 京都市ベンチャー企業目利き委員会Aランク企業認定(2013)
- 第1回京信・地域の起業家大賞(優秀賞)受賞
- 第26回中小企業優秀新技術・新製品賞(優秀賞・環境貢献特別賞)受賞
- ASIAN ENTREPRENEUR SHIP AWARD 2014(Intel Award)受賞
- 第6回ものづくり日本大賞(特別賞)受賞
- 三菱東京UFJ銀行 第4回 Rise Up Festa 最優秀賞受賞
- 三菱UFJ技術育成財団より表彰(2017)
